Insulin resistance is a key factor behind many metabolic problems, including type 2 diabetes, obesity, and fatty liver disease. Here’s a clear breakdown of what happens in the body, what causes it, and how it’s managed.
Pathophysiology What’s Happening Inside the Body
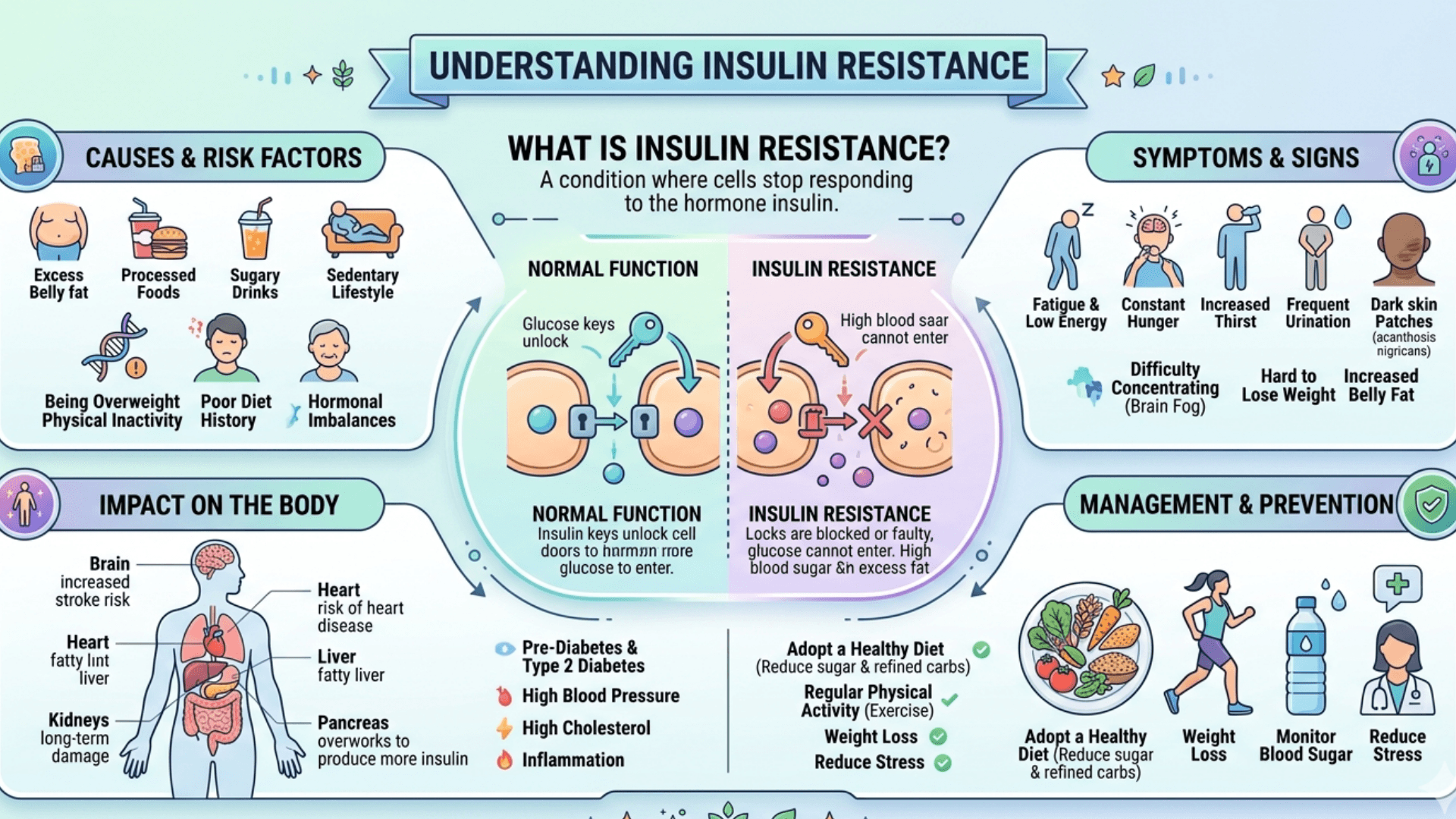
Insulin resistance means your body’s cells especially in your muscles, liver, and fat don’t respond as well to insulin as they should. Because of that, your body needs to make more insulin to keep blood sugar levels normal.
Normally, insulin helps move glucose (sugar) into cells for energy and tells the liver to slow down glucose production. When this process breaks down, muscles take up less glucose, fat tissue releases more fatty acids, and the liver keeps pumping out sugar all leading to higher blood glucose and insulin levels.
What causes this breakdown in signaling?
- Damaged insulin signaling pathways. The key proteins involved in the insulin pathway stop working efficiently, blocking normal glucose uptake.
- Fat buildup in the wrong places. When fat accumulates in the liver or muscles (not just under the skin), it interferes with insulin’s action a process called Lipotoxicity.
- Mitochondrial issues. These “power plants” of the cell may not produce energy effectively, further impairing how the body handles glucose and fat.
- Chronic inflammation. Stressed fat tissue releases inflammatory chemicals (like TNF-α and IL-6) and attracts immune cells, which worsen insulin resistance throughout the body.
How it affects different organs
- Muscles: They use most of the glucose after a meal. With insulin resistance, they can’t take up enough sugar, so blood glucose rises.
- Liver: Insulin normally stops the liver from making glucose. When the liver becomes resistant, it overproduces glucose and stores more fat, creating a cycle of worsening insulin resistance.
- Fat tissue: Unhealthy fat cells leak fatty acids into the bloodstream and send inflammatory signals that disrupt insulin’s work in other tissues.
What Causes Insulin Resistance
There’s rarely just one cause. It’s usually a mix of lifestyle, genetics, and hormonal factors.
- Excess belly fat
Visceral (deep abdominal) fat is the biggest modifiable risk factor. It’s metabolically active and releases inflammatory compounds that interfere with insulin action. - Inactivity and low muscle mass
Exercise boosts insulin sensitivity by improving how muscles use glucose. A sedentary lifestyle does the opposite. - Unhealthy diet
Diets high in refined carbs, added sugars, and saturated fats promote weight gain and fat buildup in the liver and muscles. - Genetics
Some people are more prone to insulin resistance, especially when combined with weight gain or poor diet. - Hormonal or medical conditions
Disorders like PCOS, Cushing’s syndrome, hypothyroidism, or acromegaly can worsen insulin resistance. Certain medications such as steroids or some antipsychotics can also play a role. - Aging
With age, muscle mass tends to decline while belly fat increases. Both reduce insulin sensitivity. - Sleep problems and stress
Chronic stress and poor sleep raise cortisol and other hormones that make the body less responsive to insulin. - Other metabolic issues
Conditions like fatty liver disease and abnormal cholesterol often go hand-in-hand with insulin resistance and make it worse.
How It’s Managed
The goal is to improve how the body responds to insulin, lower blood sugar levels, and prevent diabetes or related complications. Management involves lifestyle changes first, then medication if needed.
Lifestyle Changes (the Foundation)
- Weight loss
Losing even 5–10% of your body weight can significantly improve insulin sensitivity. - Exercise
Combine cardio (like brisk walking, swimming, or cycling) with strength training. Aim for at least 150 minutes of moderate activity a week, plus resistance exercises twice a week. - Healthy eating
Focus on whole, unprocessed foods. A Mediterranean-style diet — rich in vegetables, lean proteins, whole grains, and healthy fats has strong evidence for improving metabolic health. - Sleep and habits
Get enough quality sleep, limit alcohol, and avoid smoking. All three affect how the body regulates glucose and insulin.
Medications (When Needed)
If lifestyle changes aren’t enough, medications can help improve insulin sensitivity or control blood sugar.
- Metformin
Often the first choice. It reduces sugar production in the liver and slightly improves insulin sensitivity. - Thiazolidinediones (e.g., pioglitazone)
These drugs help fat cells work better and shift fat storage away from the liver and muscles. They’re effective but can cause weight gain or fluid retention. - GLP-1 receptor agonists (e.g., semaglutide, liraglutide)
These medications help with blood sugar control and appetite, often leading to significant weight loss which further improves insulin resistance. - Other medications
- SGLT2 inhibitors help the body excrete extra glucose through urine and improve heart and kidney health.
- DPP-4 inhibitors modestly lower blood sugar without weight gain.
Medication choice depends on the person’s blood sugar, weight goals, other health issues, and preferences.
When to Start Medication
For people with prediabetes, lifestyle change comes first. Metformin is considered if those changes aren’t enough especially in people with a high BMI, younger age, or a history of gestational diabetes.
Monitoring Progress
Doctors usually track weight, waist size, blood pressure, fasting glucose, HbA1c, and cholesterol. Follow-ups are done every 3–12 months depending on the situation. Regular feedback and structured programs make lifestyle changes more sustainable.
Summary
- Insulin resistance means the body’s cells don’t respond properly to insulin, mainly due to fat buildup, inflammation, and energy imbalance.
- Belly fat and inactivity are the biggest modifiable causes.
- Weight loss and regular exercise remain the most powerful ways to reverse insulin resistance.
- Medications like Metformin or GLP-1 agonists can help when lifestyle measures alone aren’t enough.
Disclaimer: The information provided in this blog post is for educational and informational purposes only and should not be considered medical advice. It is not intended to replace professional medical consultation, diagnosis, or treatment. Always seek the guidance of a qualified healthcare professional regarding any medical condition or health-related concerns. The author and publisher are not responsible for any actions taken based on the information presented in this article.